KILR Cytotoxicity Product Solutions
Specifically Measure Direct Target Cell Death
Cytotoxicity assays are required to measure target cell death via several mechanisms of action (MOA). These MOAs include antibody-dependent cell-mediated cytotoxicity (ADCC), complement dependent cytotoxicity (CDC), antibody-dependent cell phagocytosis (ADCP), cytotoxic T-cell lymphocyte mediated death (CTL), bi-specific antibody-mediated T-cell redirection, chimeric antigen receptor T-cell (CAR-T), and adoptive T-cell therapies. Such assays are designed to capture a therapeutics’ ability to cause targeted cell death via one or more primary MOAs, and to predict or mimic a physiological response. Cancer immunotherapy (immune-oncology) therapeutics are designed to treat cancers by boosting the body’s immune system to fight and kill the cancer cells. Assays for these drugs need to detect target cancer cell death when co-cultured with immune effector cells or in the presence of the complement system.
Commonly used assays for measuring cytotoxicity include reporter gene and fluorescent dye-based assays. However, these assays suffer from disadvantages, including variability induced by effector cells. Reporter gene assays only predict the MOA potential and typically require a bridging assay (as needed for ADCC) to quantify target cell death. Fluorescent and radioactive dye-based assays pose safety challenges and run the risk of the spontaneous release of dyes from the cells, causing leaks, thus impacting the accuracy and reproducibility of the data. Effector cells such as peripheral blood mononuclear cells (PBMCs) used in these assays tend to have inherent donor variability, reducing their consistency and reproducibility in lot release assays.
Eurofins DiscoverX® KILR® (Killing Immune-Lysis Reaction) cytotoxicity assay platform provides a simple, non-radioactive, and dye-free method to specifically measure target cell death that avoids the drawbacks of the other commonly used cytotoxicity assays. This homogeneous, plate-based assay platform has broad applications from screening to QC lot-release testing, particularly for immuno-oncology drug development applications such as ADCC, ADCP, CDC, CAR-T, and T-cell redirection. The flexibility of the KILR platform allows you to utilize stable cell lines or cell pools in relevant tumor models. To provide additional flexibility and ease-of-use, accelerate your drug development programs by several months with ready-to-use bioassay format, or eliminate donor-to-donor variability observed with PBMCs with the KILR CD16 Effector Cells.
Product Highlights
- Ultra-specific & Sensitive – Detect cytotoxicity only from lysed target cells and as few as 75 dead cells with high reproducibility
- Ultimate Flexibility – Ability to use with different effector cell types and run cytotoxicity assays from 30 minutes to 72 hours
- Biologically Relevant – Reflective of the true MOA of the therapeutic or ligand
- Multiple Applications – Analyze ADCC, ADCP, CDC, CAR-T, ADC, CTL mediated death, bi-specific antibody-mediated T-cell redirection, and adoptive T-cell therapies
Choose from a list of different products offered within the KILR platform to support your immuno-oncology drug discovery and development journey. These products include bioassay kits, effector cells, stable cell lines, cell pools, and retroparticles for use in screening applications and relative potency testing in lot-release programs. Consider ready-to-use bioassays with different immune effectors cells like PBMCs, NK cells, or KILR Effector Cells for potency or lot-release testing. Use KILR Effector Cells in any ADCC assay to measure target cell death. With stable cell lines and cell pools, select the relevant tumor model and study the Fc-mediated effector functions of your therapeutic antibody. Or, generate your own stable KILR cell pools/lines with retroviral particles that can transduce almost any cell line.
To obtain custom KILR cell lines, cell pools, or assays, contact custom development capabilities.
| Product | Configuration | Cat. No. |
|---|---|---|
| KILR® NCI-N87 ADCC Bioassay Kit | 2-Plate | 97-1004Y021-00173 |
| KILR® NCI-N87 ADCC Bioassay Kit | 10-Plate | 97-1004Y021-00174 |
| KILR® Raji ADCC Bioassay Kit | 2-Plate | 97-1012Y026-00169 |
| KILR® Raji ADCC Bioassay Kit | 10-Plate | 97-1012Y026-00170 |
| KILR® Raji ADCP Bioassay Kit | 2-Plate | 97-1012Y026-00179 |
| KILR® Raji ADCP Bioassay Kit | 10-Plate | 97-1012Y026-00180 |
| KILR® Daudi ADCC Bioassay Kit | 2-Plate | 97-1009Y025-00171 |
| KILR® Daudi ADCC Bioassay Kit | 10-Plate | 97-1009Y025-00172 |
| KILR® Daudi ADCP Bioassay Kit | 2-Plate | 97-1009Y025-00177 |
| KILR® Daudi ADCP Bioassay Kit | 10-Plate | 97-1009Y025-00178 |
Request qualification data set for KILR® Raji ADCC Bioassay Kit using rituximab.
| Product | Configuration | Cat. No. | |
|---|---|---|---|
| KILR® CD16 Effector Cells | 1 Vial | 97-0007-01 | |
| KILR® CD16 Effector Cells | 5 Vials | 97-0007-05 |
| Product | Configuration | Cat. No. | |
|---|---|---|---|
| KILR® ARH-77 Cell Line | Stable Cell Lines | 97-1001C017 | |
| KILR® SKBR3 Cell Pool | Cell Pool | 97-1002P018 | |
| KILR® H322 Cell Pool | Cell Pool | 97-1003P020 | |
| KILR® NCI-N87 Cell Pool | Cell Pool | 97-1004P021 | |
| KILR® A549 Cell Pool | Cell Pool | 97-1005P015 | |
| KILR® Ramos Cell Pool | Cell Pool | 97-1006P022 | |
| KILR® SKOV3 Cell Pool | Cell Pool | 97-1007P023 | |
| KILR® NCI-H292 Cell Pool | Cell Pool | 97-1008P024 | |
| KILR® Daudi Cell Pool | Cell Pool | 97-1009P025 | |
| KILR® U2OS-EGFR Cell Line | Stable Cell Lines | 97-1010C003 | |
| KILR® THP-1 Cell Pool | Cell Pool | 97-1011P014 | |
| KILR® Raji Cell Pool | Cell Pool | 97-1012P026 | |
| KILR® WIL2-S Cell Pool | Cell Pool | 97-1013P027 | |
| KILR® MCF7 Cell Pool | Cell Pool | 97-1014P028 | |
| KILR® MOLT-4 Cell Pool | Cell Pool | 97-1015P029 | |
| KILR® SK-MEL-28 Cell Pool | Cell Pool | 97-1017P031 | |
| KILR® U118-MG Cell Pool | Cell Pool | 97-1018P032 | |
| KILR® EL4 Cell Pool | Cell Pool | 97-1019P033 | |
| KILR® Jurkat Cell Pool | Cell Pool | 97-1020P019 | |
| KILR® A498 Cell Pool | Cell Pool | 97-1021P034 | |
| KILR® MDA-MB-231 Cell Pool | Cell Pool | 97-1023P036 | |
| KILR® Hut78 Cell Pool | Cell Pool | 97-1024P037 | |
| KILR® T2 Cell Pool | Cell Pool | 97-1025P038 | |
| KILR® RPMI 8226 Cell Pool | Cell Pool | 97-1026P039 | |
| KILR® CCRF-CEM Cell Pool | Cell Pool | 97-1027P040 | |
| KILR® 4T1 Cell Pool | Cell Pool | 97-1028P041 | |
| KILR® U2OS-PD-L1 Cell Line | Stable Cell Lines | 97-1029C003 | |
| KILR® K562 Cell Pool | Cell Pool | 97-1030P042 | |
| KILR® HT-1080 Cell Pool | Cell Pool | 97-1031P043 | |
| KILR® HepG2 Cell Pool | Cell Pool | 97-1032P044 | |
| KILR® SK-MEL-5 Cell Pool | Cell Pool | 97-1033P030 | |
| KILR® COLO-205 Cell Pool | Cell Pool | 97-1034P045 | |
| KILR® U2OS PD-L2 Cell Line | Stable Cell Lines | 97-1036C003 | |
| KILR® Jurkat PD-1 Cell Line | Stable Cell Lines | 97-1037C019 | |
| KILR® Jurkat LAG3 Cell Line | Stable Cell Lines | 97-1038C019 | |
| KILR® HL-60 Cell Pool | Cell Pool | 97-1039P046 | |
| KILR® HCT-116 Cell Pool | Cell Pool | 97-1040P047 | |
| KILR® BT-474 Cell Pool | Cell Pool | 97-1042P049 | |
| KILR® DU-145 Cell Pool | Cell Pool | 97-1043P050 | |
| KILR® PANC-1 Cell Pool | Cell Pool | 97-1044P051 | |
| KILR® MM-1R Cell Pool | Cell Pool | 97-1045P052 | |
| KILR® Jurkat TIM3 Cell Line | Stable Cell Lines | 97-1046C019 | |
| KILR® SR Cell Pool | Cell Pool | 97-1047P053 |
| Product | Configuration | Cat. No. |
|---|---|---|
| KILR® Retroparticles for Adherent Cells (G418) | 4 Vials x 0.5 mL | 97-0003 |
| KILR® Retroparticles for Adherent & Suspension Cells (G418) | 2 Vials x 0.5 mL (adherent cells), 2 Vials x 0.5 mL (suspension cells) | 97-0004 |
| KILR® Retroparticles for Adherent Cells (Hygromycin B) | 4 Vials x 0.5 mL | 97-0005 |
| KILR® Retroparticles for Suspension Cells (Hygromycin B) | 4 Vials x 1.0 mL | 97-0006 |
| KILR® Retroparticles for Suspension & Adherent Cells (Hygromycin B) | 2 Vials x 0.5 mL (adherent cells), 2 Vials x 0.5 mL (suspension cells) | 97-0008 |
| KILR® Retroparticles for Suspension Cells (G418) | 4 Vials x 1.0 mL | 97-0002 |
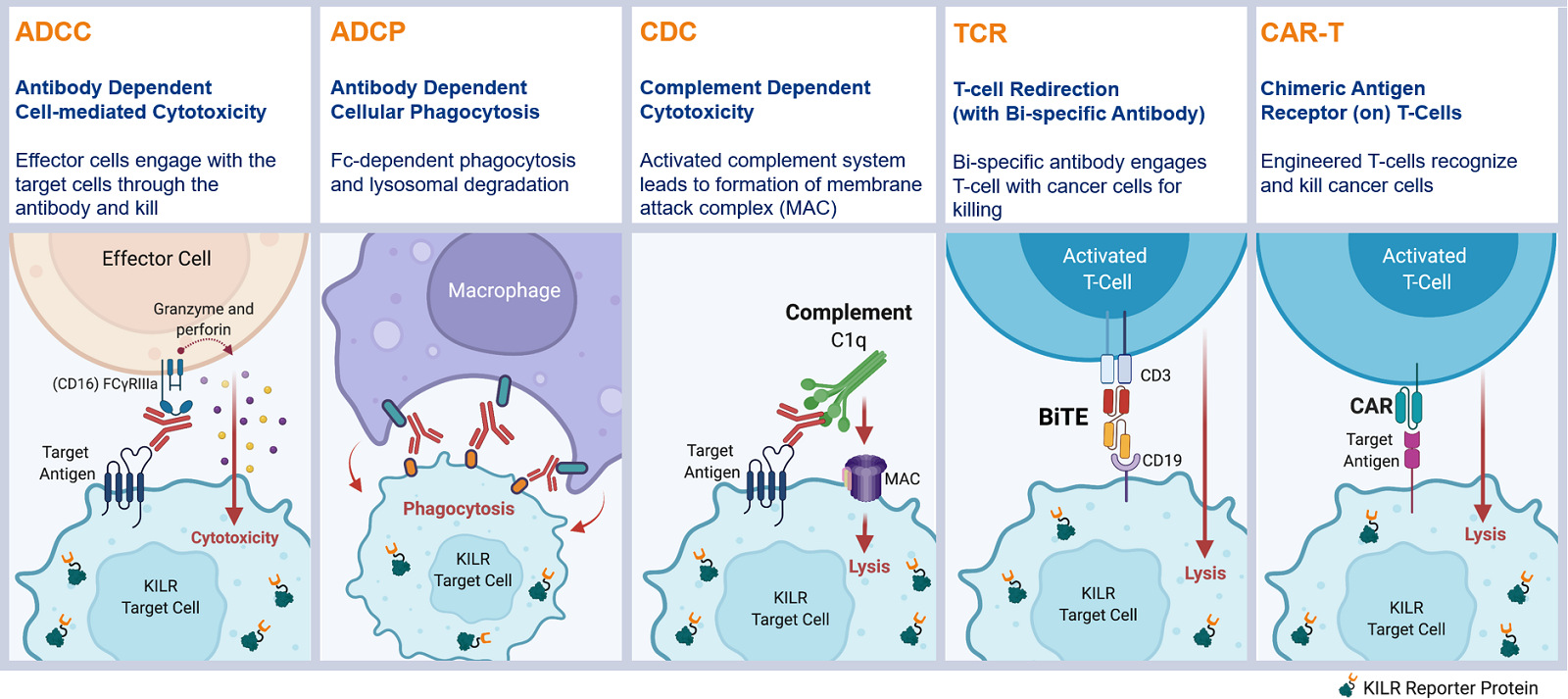
The KILR cytotoxicity assay platform specifically measures the killing of antigen-expressing target cells in co-culture with effector cells. This robust and flexible platform can be used for the quantitation of direct cell death mediated through multiple MOAs such as ADCC, ADCP, CDC, T-cell redirection, and CAR-T.
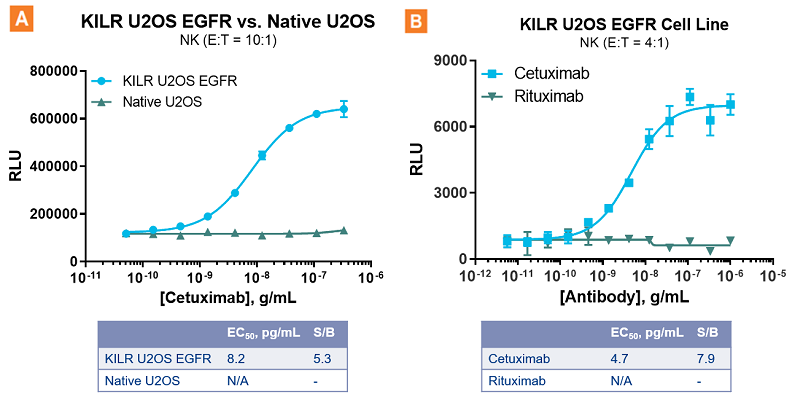
The KILR ADCC assay response is highly specific. A. Cetuximab mediates an ADCC response in U2OS cells over-expressing EGFR, but not in native U2OS cells that have low levels of endogenous EGFR. B. A highly specific ADCC response is observed with Cetuximab targeting U2OS cells expressing EGFR as opposed to that of Rituximab that targets CD20 that results in no ADCC. NK = Natural Killer. E:T = Effector Cells:Target Cells. S/B = signal-to-background.
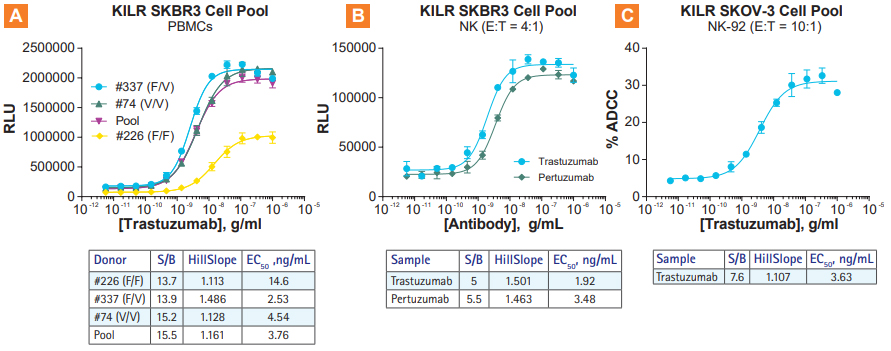
KILR ADCC Assays are compatible with multiple effector types. A. ADCC using primary PBMCs from 3 different donors in the KILR SKBR3 model B. ADCC with two HER2-targeting antibodies using primary NK cells in the KILR SKBR3 model. C. ADCC with Trastuzumab using and engineered NK-92 cell line as effector cells, with the NK-92-resistant SKOV3 cell pool. NK = Natural Killer. E:T = Effector Cells:Target Cells. S/B = signal-to-background.
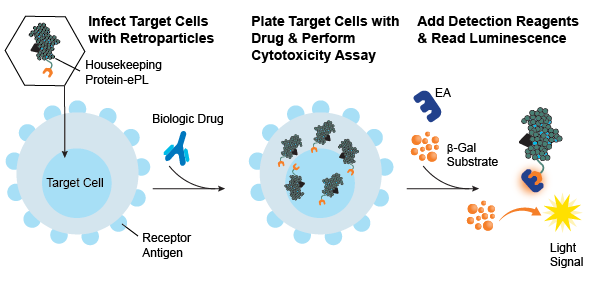
Generate cell-based cytotoxicity assays in any dividing cell type. Using Enzyme Fragment Complementation (EFC), which is based on a split β-galactosidase (β-gal) enzyme you can create your own cytotoxicity assays. Target cells expressing the receptor antigen of choice can be engineered to stably express a protein (housekeeping protein) tagged with the small β-gal enzyme donor (called the enhanced ProLabel (ePL) or β-gal reporter fragment) using KILR Retroparticles. When the stable target cell line is used in an EFC-based cytotoxicity assay (e.g., ADCP, ADCC, CDC), the EFC reaction can be measured with the addition of the large β-gal enzyme acceptor (EA) fragment that creates the active β-gal enzyme. With the addition of hydrolyzing substrate, a chemiluminescent output can be detected on any bench top luminometer.
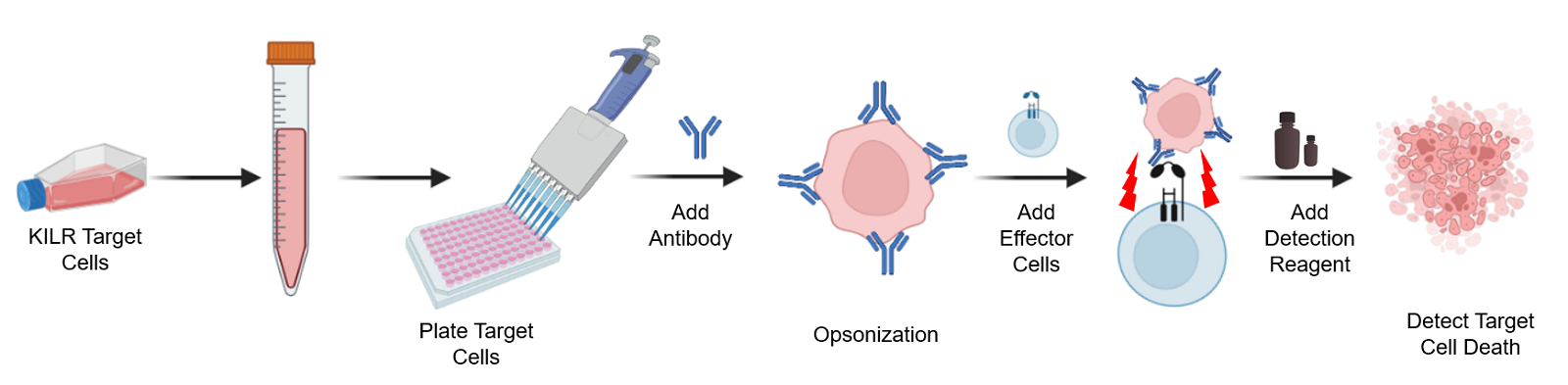
The KILR ADCC assay workflow. KILR target cells of interest are engineered to stably express a housekeeping protein that is tagged with a reporter fragment (KILR reporter protein). These cells are plated, followed by the addition of the test antibody to the target cells. Following opsonization, effector cells (e.g. KILR CD16 Effector Cells) are added to the wells containing the target cells. Effector-mediated killing releases the KILR reporter protein into the media that is detected by the addition of detection reagents and results in a chemiluminescent signal that can be read on any standard benchtop luminometer.
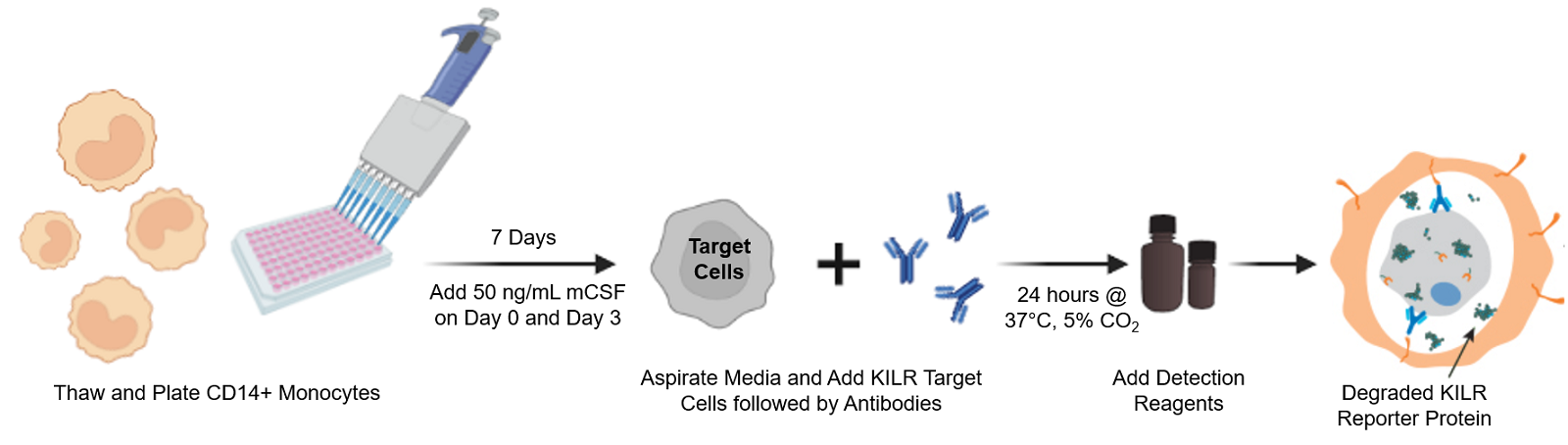
The KILR ADCP assay workflow. CD14+ Monocytes are thawed and plated with the addition of 50 ng/mL of mCSF. Following 7 days of incubation, KILR target cells of interest, expressing the KILR reporter protein and a test antibody, are added to the plate wells containing the monocytes, now differentiated into macrophages. The macrophages phagocytose the opsonized KILR target cells leading to the degradation of the KILR reporter protein. The addition of detection reagent causes cell lysis. Complementation of two β-gal fragments with the addition of substrate results in a dose-dependent decrease in chemiluminescent signal with increasing concentrations of the test antibody. Note a shorter protocol is available and included within the ADCP application note.
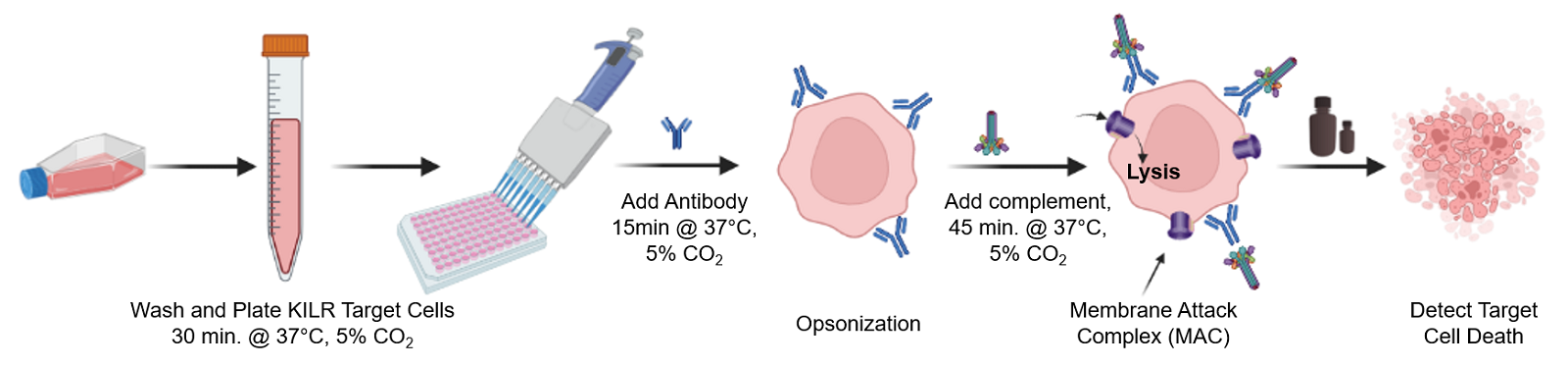
The KILR CDC assay workflow. KILR target cells of interest are engineered to stably express a housekeeping protein that is tagged with a reporter fragment (KILR reporter protein). These cells are plated followed by addition of test antibody to target cells. Following opsonization, appropriate complement is added to the wells containing the target cells. Complement-mediated killing releases the KILR reporter protein into the media, which is detected by the addition of detection reagents and results in a chemiluminescent signal that can be read on any standard bench top luminometer.
The KILR platform is based on the industry-validated Enzyme-Fragment Complementation (EFC) technology. EFC is based on two recombinant β-galactosidase (β-gal) enzyme fragments that act as an enzyme acceptor (EA) and an enzyme donor (ED). Separately, the fragments are inactive, but when combined, they form an active β-gal enzyme that hydrolyzes its substrate to produce a chemiluminescence signal.
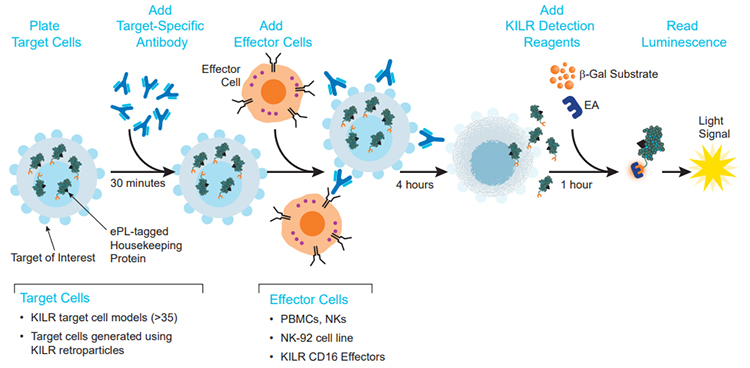
The KILR ADCC assay principle. Target cells expressing the relevant antigen are engineered to stably express a housekeeping protein tagged with a reporter β-gal ED fragment called enhanced ProLabel® (ePL). The β-gal enzyme is inactive when the reporter fragment (also called the KILR reporter protein) is not paired with its larger β-gal EA fragment. On incubation of KILR target cells with a test antibody and appropriate effector cells, effector cell-mediated killing releases the tagged protein into the media. The presence of both EA and ED fragments leads to the formation of the active β-gal enzyme that hydrolyzes the substrate to give a chemiluminescent output detected on any benchtop luminometer.
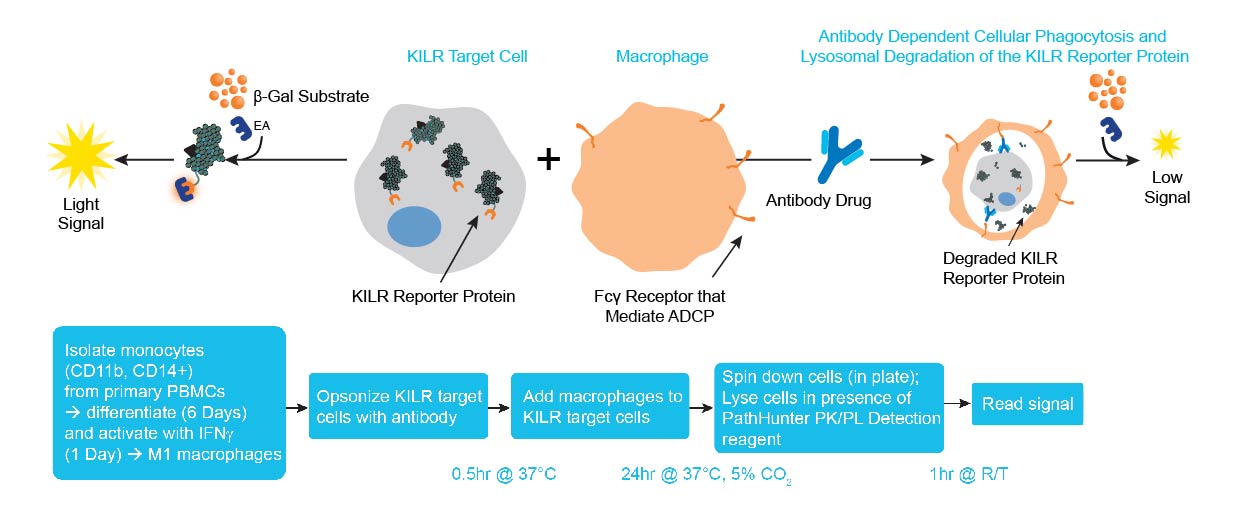
The KILR ADCP assay principle. ADCP measures antibody-dependent phagocytosis of target cells, typically mediated by primary human macrophages. KILR target cells, containing the KILR reporter protein (including the β-gal ED fragment also called ePL), undergo phagocytosis by effector cells (e.g. macrophages differentiated from monocytes) leading to lysosomal degradation of the KILR reporter protein. In this assay, the KILR target cells are opsonized with the antibody of interest, then co-cultured with the effector cells. Macrophages perform phagocytosis of the target cells. All non-phagocytosed cells are lysed and the detection reagent containing the complementing β-gal EA fragment is added to the lysed cells. Complementation of ED and EA with the addition of substrate results in the detection of the non-phagocytosed KILR protein present. A high chemiluminescent signal indicates minimum killing, while a low signal correlates to higher phagocytosis or increased ADCP activity. Note: other immune effector cells can also be used for the ADCP assay.
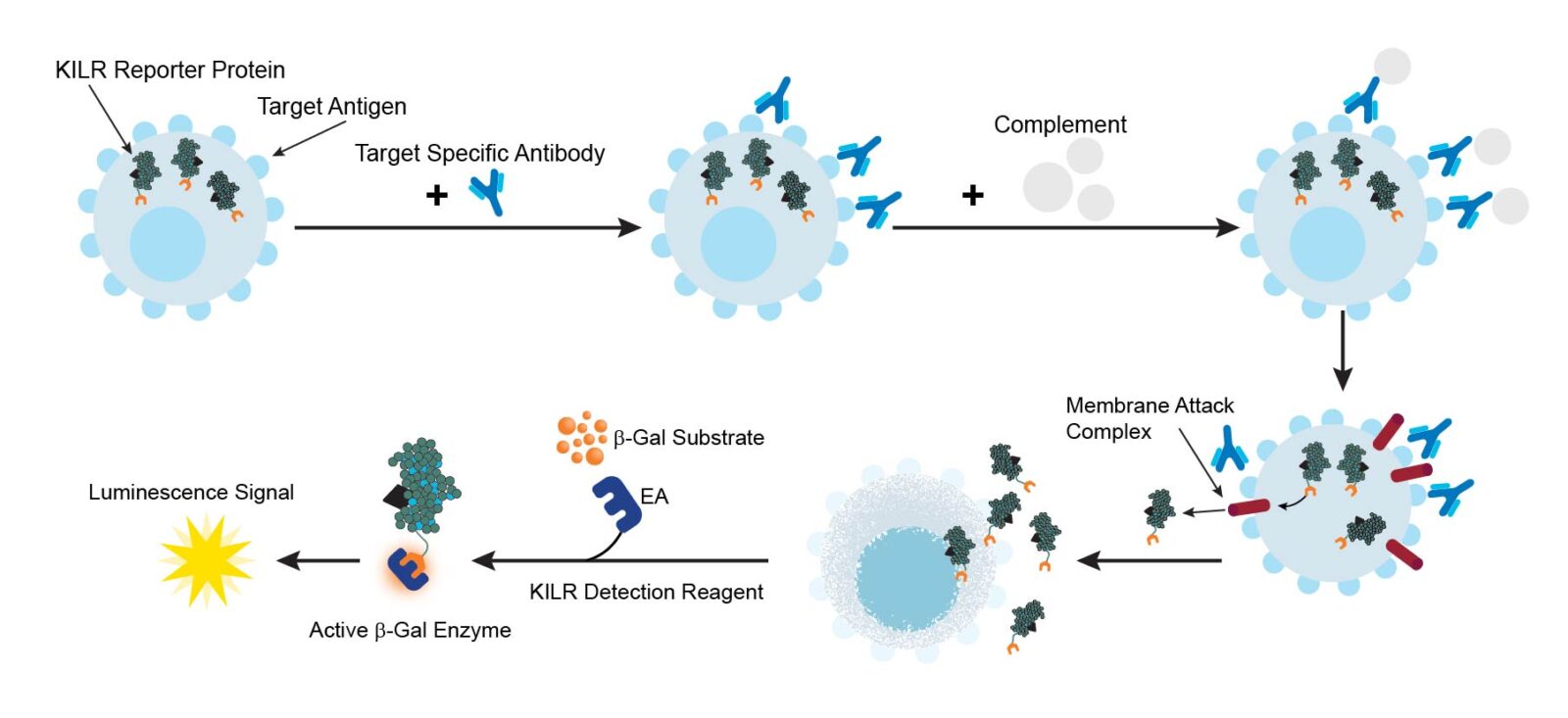
The KILR CDC assay principle. CDC is mediated by the C1 complex that initiates deposition of C3b fragments on target cells. The C3b in conjunction with C4b and C2a forms the C5 convertase enzyme that cleaves C5 into C5a and C5b. C5b then participates in the formation of the membrane attack complex or MAC (C5b-C6-C7-C8-C9). MAC is responsible for the cytolytic function of the complement system resulting in subsequent target cell lysis. The KILR reporter protein (including the β-gal ED fragment also called ePL) is stably expressed in the target cells and released into the media when these target cells undergo lysis as a result of MAC formation. The amount of total KILR reporter protein is detected by the addition of detection reagents containing the β-gal EA fragment. Complementation of ED and EA facilitates detection of the remaining amount of KILR protein present, and with the addition of subtrate, hydrolysis occurs and a chemiluminescent signal can be detected on any bench top luminometer.
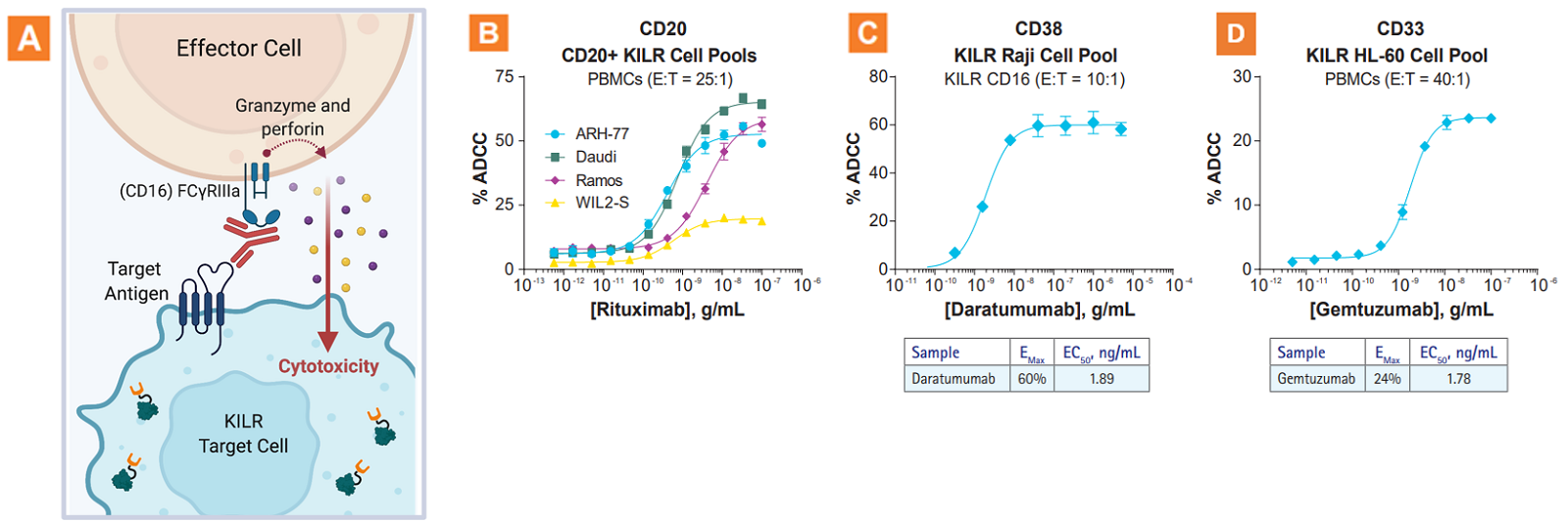
The KILR ADCC assay application. A. Graphic illustration of the KILR ADCC MOA. B. Rituximab-mediated ADCC in four different CD20+ B-lymphoblast KILR models (ARH-77, Daudi, Ramos, and WIL2-S) using primary PBMCs. C. ADCC mediated by the anti-CD38 therapeutic antibody, Daratumumab, in the KILR Raji cell model using engineered effector cells (KILR CD16 effector cells). D. ADCC mediated by the anti-CD33 therapeutic antibody, Gemtuzumab (approved for treatment of AML), in the KILR HL-60 cell model using primary human PBMCs. This data demonstrates that KILR ADCC assays can be used to evaluate diverse cancer models. E:T = Effector Cells:Target Cells.
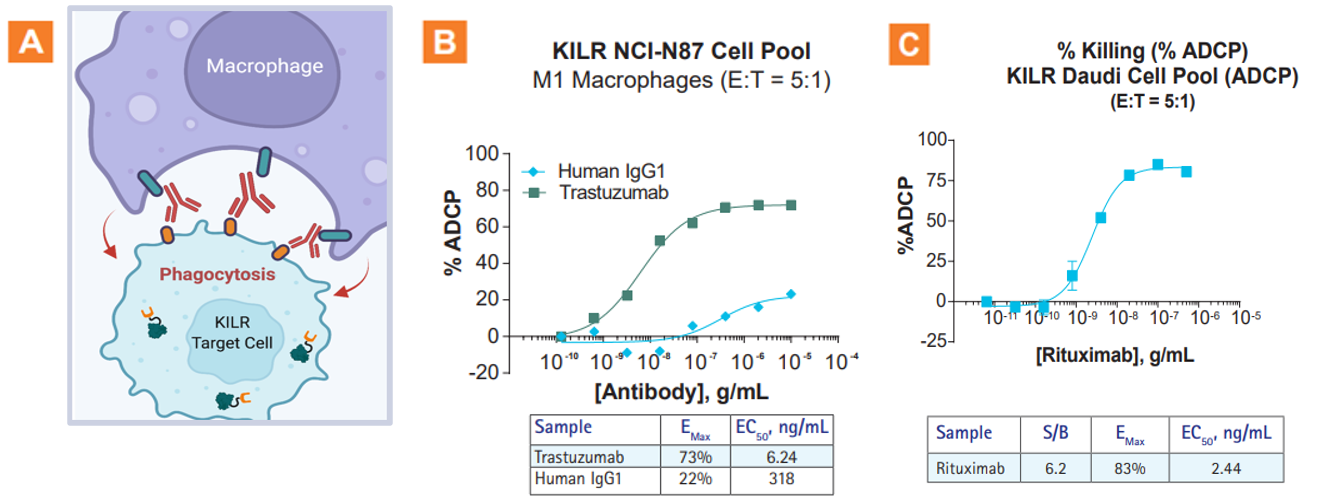
The KILR ADCP assay application. A. Graphic illustration of the KILR ADCP MOA. B. Robust ADCP observed with trastuzumab with KILR NCI-N87 cell pool (a HER2+ solid tumor model). C. A robust dose-dependent increase in % ADCP was observed with increasing concentrations of rituximab. An EMAX value of 83% was obtained with a low EC50 of 2.4 ng/mL. E:T = Effector Cells:Target Cells.
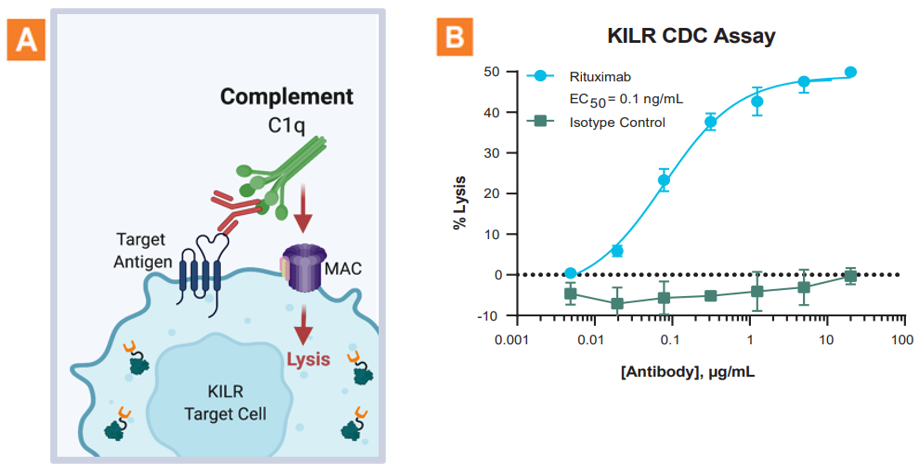
The KILR CDC assay application. A. Graphic illustration of the KILR CDC MOA. B. CDC activity was assessed with KILR Raji bioassay cells. This assay responds specifically to rituximab and exhibits a wide dynamic range as indicated by the % lysis of the KILR target cells.

The KILR T-cell redirection assay application. A. Graphic illustration of the KILR t-cell redirection MOA. BiTEs are chimeric antibodies with dual specificities that target tumor cells by harnessing the power of the immune effector cells. The BiTE molecule binds to a tumor-associated antigen such as CD19. The KILR CD16 Effector Cells, which express CD3 endogenously, rapidly kills target cells via t-cell redirection. B. KILR CD16 Effector Cells were incubated with KILR Raji cells and blinatumomab (an FDA approved BiTE molecule) for 4, 5, or 6 hours at 37°C. Optimal assay window (max killing of >60%) was observed at 6 hour incubation with KILR CD16 effectors with an EC50 of 15.5 pg/mL.
The development of antibody-based therapeutics to target the killing of tumor cells has revolutionized the space of immuno-oncology and the…
Read MoreWith the increasing industry focus on antibody drugs, there is an ever-greater need for functional bioassays that interrogate the therapeutic…
Read MoreAudrey B. Bergeron, B.A. and Hannah J. Gitschier, M.S. Corning Incorporated, Life Sciences Kennebunk, Maine
Read MoreGaurav Agrawal, Ph.D.Scientific Development Manager
Read MoreDiscoverX CASSS 2017 Presentation Presented by Dr. Jane Lamerdin, Director R&D, DiscoverX May 9, 2017
Read MoreAndrew Green Sr. Business Development Manager
Read MoreJane E. Lamerdin, Ph.D. Vice President, Research & Development Eurofins DiscoverX
Read MoreDiscoverX PEGS 2017 Presentation Presented By: Jane Lamerdin, Ph.D.
Read MoreVenkatesh Chari, Ph.D. Scientific Market Development Manager Eurofins DiscoverX Products LLC
Read MoreCell-based assays play an important role in determination of mechanism of action (MOA) for therapeutic antibodies, particularly when evaluating their…
Read MoreThe clinical success of an ever-increasing array of biologics has led to the development of a wide spectrum of immunomodulatory…
Read MoreSuccess of ADCC assays is highly dependent on the quality of effector cells used. However, human primary cells (such as…
Read MoreAntibody-dependent cell-mediated cytotoxicity (ADCC) is the mechanism by which many Class I therapeutic antibodies, including rituximab and trastuzumab, achieve clinical…
Read MoreChimeric antigen receptor (CAR)-T cells, which are engineered to recognize target cell surface antigens expressed on tumor cells, have shown…
Read MoreClass I therapeutic antibodies achieve their clinical efficacy not only by binding to their target antigen, but also through Fc…
Read MoreFollowing the recent regulatory approvals of a number of checkpoint inhibitor therapeutics, development of a wide array of immunomodulatory therapeutic…
Read MoreThe clinical success of an ever-increasing array of biologics has led to the development of a wide spectrum of immunomodulatory…
Read MoreListen to the US PEGS 2023 conference talk by Venkatesh Chari, Ph.D. on MOA-reflective, functional cell-based assays (cell lines and…
Watch NowLearn how KILR® cytotoxicity assays provide a simple, non-radioactive and dye free method to specifically measure target cell death in…
Watch NowLearn how KILR CD16 effector cells ensure assay reproducibility with higher signal-to-background ratios, without worrying about donor variability. PBMC effector…
Watch NowLearn about Eurofins DiscoverX cell line engineering, assay development, and recombinant enzyme production custom capabilities. Find out more about cell…
Watch NowComprehensive Eurofins DiscoverX product list to enable your drug discovery and development programs focused on checkpoint receptors, cytokine receptors/interleukins, cytotoxicity,…
Read MoreAccelerate your biologics development from discovery to QC lot release with cell-based assays. Comprehensive list of bioassay kits, cell lines,…
Read MoreThe webinar focusses on measuring antibody-mediated cell death using DiscoverX’s KILR Bioassays, and how they can be used to measure…
Watch NowPBMC effector cells have inherent donor variability, reducing their consistency and reproducibility in lot release assays. Antibody-Dependent Cell Cytotoxicity (ADCC)…
Watch NowInherent donor variability of immune effector cells presents a significant challenge for developing a robust and reproducible ADCC assays for…
Watch NowTherapeutic regulators require assay data on the impact of Fc effector-mediated function of antibodies during their development as therapeutics. The…
Read MoreTherapeutic antibodies that can elicit different applications of cell-mediated cytotoxicity, such as Antibody-Dependent Cell-mediated Cytotoxicity (ADCC) and many other effector…
Read MoreClass I therapeutic antibodies achieve their clinical efficacy by binding to their target antigen and through Fragment crystallisable region (Fc)…
Read MoreCell-based assays are useful for understanding cellular pathways; characterizing targets and their function; identifying, screening, and optimizing ligands or drugs…
Read MoreMost therapeutic antibodies have a Fab’ region that binds the antigen, and an Fc region which binds to and activates…
Read MoreClass I therapeutic antibodies achieve their clinical efficacy not only by binding to their target antigen but also through Fc…
Read MoreRegulatory T cells (Tregs) are indispensable for maintaining self-tolerance by suppressing conventional T cells. On the other hand, Tregs promote…
Read MoreSmall ubiquitin-like modifier (SUMO) is a member of a ubiquitin-like protein superfamily. SUMOylation is a reversible posttranslational modification that has…
Read MoreEvaluation of cytotoxicity is a critical step in the selection of CD3-bispecific antibodies (BsAbs) for cancer immunotherapy.
Read MoreRobust & Reproducible Cell-Based Assays for Potency Testing
Read MoreRobust, Ready-to-Use ADCC & ADCP Bioassays to Accelerate Your Drug Development Program The KILR® cytotoxicity assay platform developed by Eurofins…
Read MoreProducts & Custom Development Capabilities.
Read MoreDiscover valuable insights on breakthroughs, innovative strategies, and cell-based approaches for immunotherapeutic discovery and development.
Read MoreDiscover valuable insights on breakthroughs, innovative strategies, and cell-based approaches for immunotherapeutic discovery and development.
Read More
Single donor-derived human primary cells to drive robust & reproducible ADCC & T-cell redirection
Read More
Ready-to-use, cell-based bioassay kits to measure direct cell death for screening, characterization, & potency testing
Read More
Complete set of parental cell lines, vectors, kits, & retroparticles to build your own stable cell lines & cell-based assays
Read More
Custom cell lines, kits, assays, & protein development capabilities optimized to fit your requirements
Read More
